Raw Material Shortages – Midas is actively approaching these challenges
In the recent years, generic drug shortages have been raised increasing attention in the EU. The reasons for these drug shortages are manifold. They could be caused by quality issues at a key supplier, unexpected increase of demand for a certain product, price pressure leading to strategy changes/ outsourcing activities or incidents causing production stops in the chemical supply chain.
Although there might be a large number of manufacturers for a certain API, these are often directly or indirectly dependent on just a few raw material / starting material suppliers. In case of an incident at the raw material supplier, it might have an impact on the API availability in the whole market.
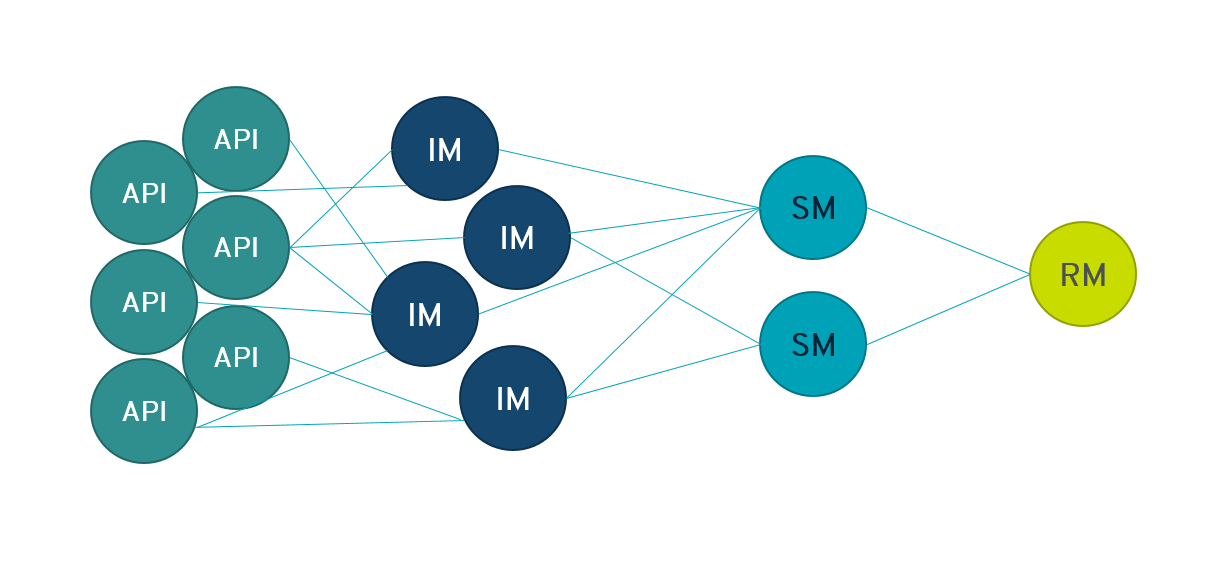
-
Examples of current events that have potential to cause API supply shortages in the EU:
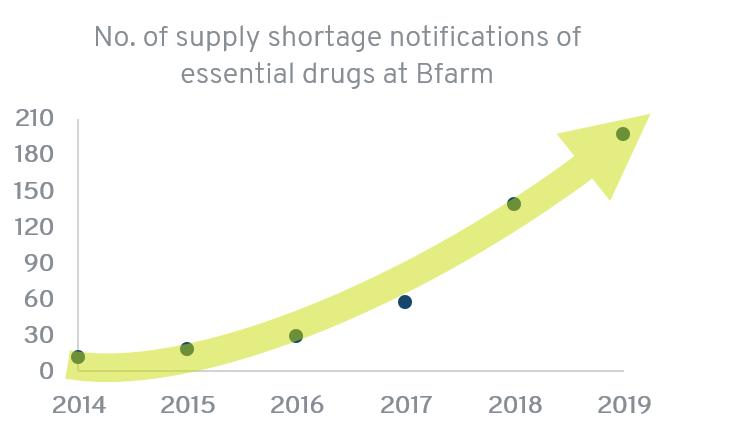
BfArM reports an increase in supply shortage notifications in the recent years (Source: German Government's comments on supply shortages, printed matter 19/13357; dossier on supply shortages of BfArM https://www.bfarm.de/EN):
- In the recent years China intensified its effective and overdue measures to improve the environmental situation. By shrinking the number of outdated production capacities and setting stricter emission limits and controls, especially capacities for raw materials based on critical chemistry are affected.
- Currently, the National Blue Sky Action Plan 2018 – 2020 is in place, targeting among other the reduction of average density of fine particulate matter (PM2.5) by 15% (vs. 2015).
- The devastating explosion in Yancheng in China's eastern Jiangsu province in March 2019, caused a special action plan, incl. e.g. the target to decrease the number of chemical producers in Jiangsu from 5,433 to 2,000 by end of 2020 and down to max. 1,000 by 2022.
- UK frequently updates a list of drugs that cannot be parallel exported from the UK because they are needed for UK patients.
- Temporary closures of manufacturing sites due to Covid-19 pandemic and the related impact on the global logistics sector.
- On March 3rd 2020 India had executed an export ban of 26 APIs and formulations to secure the availability of these drugs in the domestic market. India has lifted the export ban partially in second week of April.

- In the recent years China intensified its effective and overdue measures to improve the environmental situation. By shrinking the number of outdated production capacities and setting stricter emission limits and controls, especially capacities for raw materials based on critical chemistry are affected.
What is Midas‘ strategy to address these shortages?
- Information is crucial! We use our worldwide network of partners and Midas global affiliates to collect information about critical intermediates and about supply chain dependencies.
-
We use this information to develop solutions for de-risking of critical supply chains:
- We are cooperating with global partners (especially in Asia) to jointly transfer technologies to local manufacturing sites.
- We built up alternative sources for substances under shortage.
- We agglomerate demands for intermediates under shortage, where a development just for inhouse consumption is not justified.
-
We implement supply chain risk evaluations in our sourcing activities.
We are looking forward to an exchange about your ideas, please get in contact with us!
- Are you interested in intermediates / raw materials that are short in the market? We might be working on a solution already.
- Do you intend to minimize risks in your supply chain by adding a sustainable source from a different territory?
- Do you hold technologies of interest for the EU-market or are you based in EU and consider to re-activate a formerly stopped technology? We are open to discuss about joint transfer activities and/or a marketing cooperation.
In case of interest in this topic, please don't hesitate to contact us.
Your Contact

Dr. Danielle Leone-Stumpf
International Division
Senior Director Intermediates for Pharma & BioPharma
Rheinstr. 49
55218 Ingelheim
Germany